I realized that it's been a while since using many things (generator, lawnmower, trimmers and 1 LSX equipped van that contain regular gas and some with 2 stroke oil mixes How long does regular gas stay good to use? I likely have about 20 gals. of gas and about a gal of 2 stroke mix. in different things. What do you do with bad gas? I seem to recall some manufacture claiming there is an additive that makes old fuel useable - anybody ever hear about this?
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Good or Bad Gas
- Thread starter chromebumpers
- Start date
All gasoline breaks down over time, most gas absorbs moisture from the air to some extent, ethanol laced fuel is especially prone to this moisture absorption.
ethanol is hygroscopic
RELATED USEFUL INFO
Because ethanol has an octane rating of 113, adding 10% -25% ethanol to gasoline raises the finished fuel's octane rating, but lowers its ability to help lubricate the upper cylinder surfaces, and tends to increase the chances for corrosion
http://www.intercepteft.com/calc.html
http://www.georgebelton.com/E85Calculator.php
https://docs.google.com/a/langevins.net/file/d/0B66vrbT1HQ19YjVhOWlaOGRtWXc/edit
http://forums.turbobricks.com/showthread.php?t=73061
http://garage.grumpysperformance.co...ng-e85-in-your-old-muscle-car.2141/#post-5780
Technical facts about the mentioned fuels:
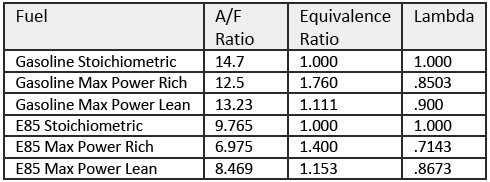
E85 requires 42% more fuel to reach stoich,
even if that is not what you may come up with when doing calculations, based on the table below.
This is because the injector flow is slightly different when using E85 among many other things.
I can´t really think of at this time (will be added at a later time).
Fuel ........................ AFRst ........ FARst ....... Equivalence Ratio ... Lambda
Gas stoich ................ 14.7 .......... 0.068 ................ 1 ................... 1
Gas max power rich .... 12.5 .......... 0.08 ................. 1.176 .............. 0.8503
Gas max power lean .... 13.23 ........ 0.0755 .............. 1.111 ............. 0.900
E85 stoich .................. 9.765 ....... 0.10235 ............ 1 ................... 1
E85 max power rich ...... 6.975 ....... 0.1434 .............. 1.40 ............... 0.7143
E85 max power lean ..... 8.4687 ...... 0.118 ............... 1.153 .............. 0.8673
E100 stoich ................ 9.0078 ...... 0.111 ............... 1 .................... 1
E100 max power rich .... 6.429 ........ 0.155 .............. 1.4 .................. 0.714
E100 max power lean .... 7.8 .... ...... 0.128 .............. 1.15 ................ 0.870
The term AFRst refers to the Air Fuel Ratio under stoichiometric, or ideal air fuel ratio mixture conditions. FARst refers to the Fuel Air Ratio under stoichiometric conditions, and is simply the reciprocal of AFRst.
Equivalence Ratio is the ratio of actual Fuel Air Ratio to Stoichiometric Fuel Air Ratio; it provides an intuitive way to express richer mixtures. Lambda is the ratio of actual Air Fuel Ratio to Stoichiometric Air Fuel Ratio; it provides an intuitive way to express leanness conditions (i.e., less fuel, less rich) mixtures of fuel and air.
When driving purely on E85 you can blend it with up to 25% gasoline in case you want to raise the AFR number used to produce max. power. In that case you can raise the boost even further since the volume of fuel needed to reach the desired lambda is decreased.
hy·gro·scop·ic
ˌhīɡrəˈskäpik/
adjective
(of a substance) tending to absorb moisture from the air.
water combined with ethanol is highly corrosive if left in contact with most metals over long time periods
the ability of gasoline to quickly and easily vaporize tends to degrade over time making it significantly less easily ignited as its the vapor not the liquid component of the fuel that burns the most efficiently, normally heat helps rapidly atomize and vaporize the fuel, but as fuel ages the easily vaporized components tend to be lost to evaporation, leaving a less easily ignited and usually DARKER COLORED LIQUID.
mixing the remaining old gas with new fuel will generally help but not cure the issue!
I use STA-BIL marine fuel additive in my lesser used engines, but reading the info on various site the LONGEST listed time frame it seems to work for as designed was about 2 years, if I had gas over 2 years old ID personally drain the tank, add stabil, plus a name brand fuel system cleaner and a fresh fuel filter,add fresh non-alcohol laced fuel, and flush the fuel rail and or car fuel bowls, before using the car or engine,
Id probably use the old fuel I drained to clean parts or ignite a trash pile, yeah you might be able to mix it with fresh fuel and use it but the chances of it having absorbed moisture, separated into various less than ideal for use as a fuel,components while sitting for over two years,or causing issues with the engine that might damage it,would just not be worth the risk in my opinion.

http://www.goldeagle.com/tips-tools/top ... s-debunked
http://www.sta-bil360.com/how-it-works
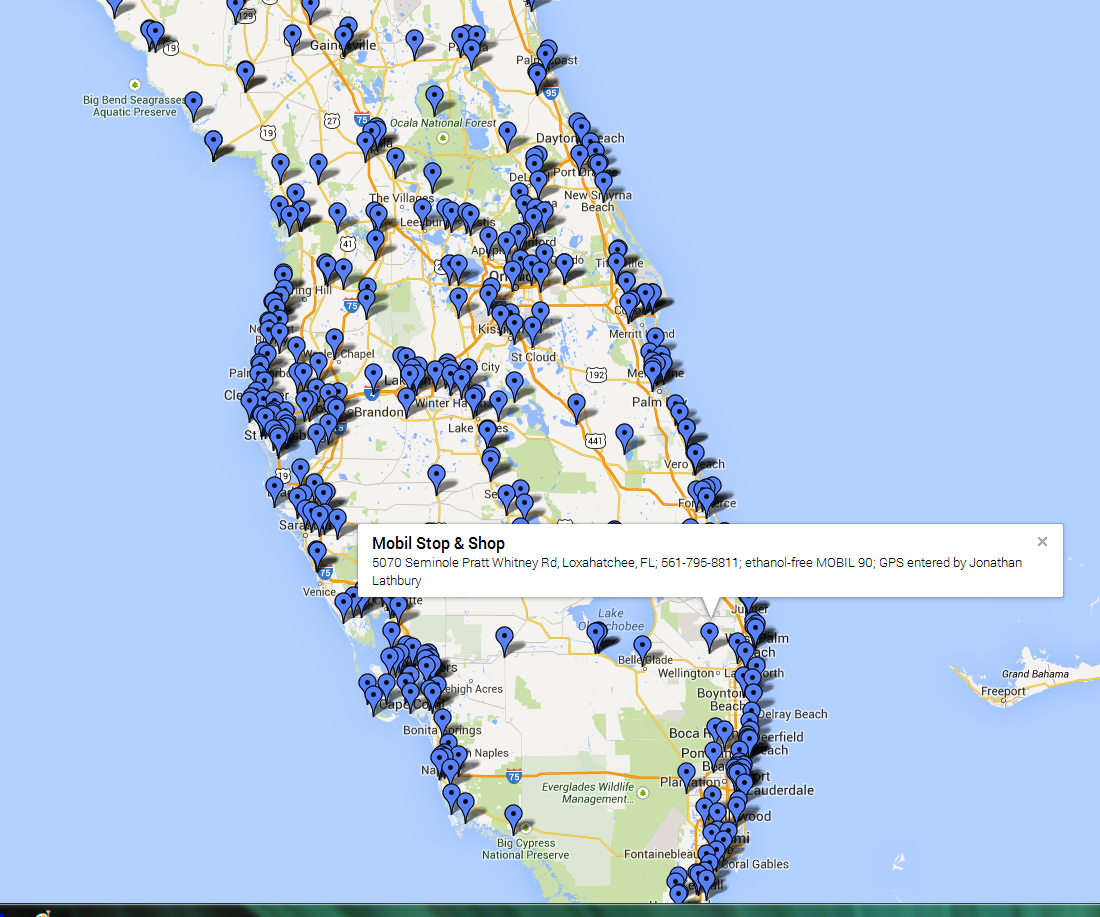
there ARE ETHANOL FREE GAS STATIONS LISTED, when you NEED TO FILL A MUSCLE CAR TANK, HERES MY LOCAL STATION, yes it costs more short term, easily an extra 70-80 cents a gallon, but you don,t run nearly the same risk of trashing your muscle cars engine and fuel delivery systems
http://pure-gas.org/extensions/map.html
no IF your using e85 you just need a ignition advance curve thats constantly modified with a knock sensors input,

http://injectordynamics.com/articles/e85/
https://www.enginelabs.com/engine-t...t-e85-and-ethanol-16-things-you-need-to-know/
https://www.6speedonline.com/forums...hot-cold-e85-vs-gas-theoretical-analysis.html
http://garage.grumpysperformance.co...octane-for-compression-ratio.2718/#post-86754
http://garage.grumpysperformance.co...ne-running-a-turbo-s-on-e85.10990/#post-73199
https://www.onallcylinders.com/2015...-is-safe-effective-compression-ratio-for-e85/
the most effective compression ratio for gas , is in the 10:1-12.7:1 range (depends on true octane and f/a ratio and several other factors)
the most effective compression ratio for e85 ethanol , is in the 12:1-15:1 range (depends on true octane and f/a ratio and several other factors)
E85 PRO’S
ethanol is hygroscopic
RELATED USEFUL INFO
Because ethanol has an octane rating of 113, adding 10% -25% ethanol to gasoline raises the finished fuel's octane rating, but lowers its ability to help lubricate the upper cylinder surfaces, and tends to increase the chances for corrosion
http://www.intercepteft.com/calc.html
http://www.georgebelton.com/E85Calculator.php
https://docs.google.com/a/langevins.net/file/d/0B66vrbT1HQ19YjVhOWlaOGRtWXc/edit
http://forums.turbobricks.com/showthread.php?t=73061
http://garage.grumpysperformance.co...ng-e85-in-your-old-muscle-car.2141/#post-5780
Transporting Limited Amounts Of Fuel, Or Waste Oil
I don,t know how many of you gentlemen have shop welder/generators or other lawn mowers or similar equipment that requires you to buy and transport a bit of fuel for thier use, but Ive noticed that finding a well designed structually strong 5 gallon fuel transport container thats rather leak and...
garage.grumpysperformance.com
E85 requires 42% more fuel to reach stoich,
even if that is not what you may come up with when doing calculations, based on the table below.
This is because the injector flow is slightly different when using E85 among many other things.
I can´t really think of at this time (will be added at a later time).
Fuel ........................ AFRst ........ FARst ....... Equivalence Ratio ... Lambda
Gas stoich ................ 14.7 .......... 0.068 ................ 1 ................... 1
Gas max power rich .... 12.5 .......... 0.08 ................. 1.176 .............. 0.8503
Gas max power lean .... 13.23 ........ 0.0755 .............. 1.111 ............. 0.900
E85 stoich .................. 9.765 ....... 0.10235 ............ 1 ................... 1
E85 max power rich ...... 6.975 ....... 0.1434 .............. 1.40 ............... 0.7143
E85 max power lean ..... 8.4687 ...... 0.118 ............... 1.153 .............. 0.8673
E100 stoich ................ 9.0078 ...... 0.111 ............... 1 .................... 1
E100 max power rich .... 6.429 ........ 0.155 .............. 1.4 .................. 0.714
E100 max power lean .... 7.8 .... ...... 0.128 .............. 1.15 ................ 0.870
The term AFRst refers to the Air Fuel Ratio under stoichiometric, or ideal air fuel ratio mixture conditions. FARst refers to the Fuel Air Ratio under stoichiometric conditions, and is simply the reciprocal of AFRst.
Equivalence Ratio is the ratio of actual Fuel Air Ratio to Stoichiometric Fuel Air Ratio; it provides an intuitive way to express richer mixtures. Lambda is the ratio of actual Air Fuel Ratio to Stoichiometric Air Fuel Ratio; it provides an intuitive way to express leanness conditions (i.e., less fuel, less rich) mixtures of fuel and air.
When driving purely on E85 you can blend it with up to 25% gasoline in case you want to raise the AFR number used to produce max. power. In that case you can raise the boost even further since the volume of fuel needed to reach the desired lambda is decreased.
hy·gro·scop·ic
ˌhīɡrəˈskäpik/
adjective
(of a substance) tending to absorb moisture from the air.
water combined with ethanol is highly corrosive if left in contact with most metals over long time periods
the ability of gasoline to quickly and easily vaporize tends to degrade over time making it significantly less easily ignited as its the vapor not the liquid component of the fuel that burns the most efficiently, normally heat helps rapidly atomize and vaporize the fuel, but as fuel ages the easily vaporized components tend to be lost to evaporation, leaving a less easily ignited and usually DARKER COLORED LIQUID.
mixing the remaining old gas with new fuel will generally help but not cure the issue!
I use STA-BIL marine fuel additive in my lesser used engines, but reading the info on various site the LONGEST listed time frame it seems to work for as designed was about 2 years, if I had gas over 2 years old ID personally drain the tank, add stabil, plus a name brand fuel system cleaner and a fresh fuel filter,add fresh non-alcohol laced fuel, and flush the fuel rail and or car fuel bowls, before using the car or engine,
Id probably use the old fuel I drained to clean parts or ignite a trash pile, yeah you might be able to mix it with fresh fuel and use it but the chances of it having absorbed moisture, separated into various less than ideal for use as a fuel,components while sitting for over two years,or causing issues with the engine that might damage it,would just not be worth the risk in my opinion.

http://www.goldeagle.com/tips-tools/top ... s-debunked
http://www.sta-bil360.com/how-it-works
there ARE ETHANOL FREE GAS STATIONS LISTED, when you NEED TO FILL A MUSCLE CAR TANK, HERES MY LOCAL STATION, yes it costs more short term, easily an extra 70-80 cents a gallon, but you don,t run nearly the same risk of trashing your muscle cars engine and fuel delivery systems
http://pure-gas.org/extensions/map.html

GRUMPY... Do you think a water/meth setup would be benificial to my engine since it is fueled by E-85?
no IF your using e85 you just need a ignition advance curve thats constantly modified with a knock sensors input,


http://injectordynamics.com/articles/e85/
https://www.enginelabs.com/engine-t...t-e85-and-ethanol-16-things-you-need-to-know/
https://www.6speedonline.com/forums...hot-cold-e85-vs-gas-theoretical-analysis.html
http://garage.grumpysperformance.co...octane-for-compression-ratio.2718/#post-86754
http://garage.grumpysperformance.co...ne-running-a-turbo-s-on-e85.10990/#post-73199
https://www.onallcylinders.com/2015...-is-safe-effective-compression-ratio-for-e85/
the most effective compression ratio for gas , is in the 10:1-12.7:1 range (depends on true octane and f/a ratio and several other factors)
the most effective compression ratio for e85 ethanol , is in the 12:1-15:1 range (depends on true octane and f/a ratio and several other factors)

Advantages of Ethanol-Enriched Racing Fuels
HORSEPOWER:
Because Ethanol contains oxygen, it has a very low power stoichiometric
when compared to gasoline fuels (6.5 compared to 12.5). Ethanol must be run at much richer
mixtures than gasoline, more than offsetting the lower energy per unit volume. The net
energy released per cycle is higher and this results in more horsepower.
For example, if a pound of gasoline is burned at its preferred max power air fuel mixture of
12.5/1, it will release approximately 19,000 BTU’s of energy, where ethanol run at its preferred
power stoichiometric of 6.5/1 will release approximately 24,400 BTU’s. By comparison,
methanol releases slightly more, about 27,650 BTU’s. The more ethanol there is in gasoline, the
more powerful it is as a motor fuel. Typically, you can expect at least 5% more horsepower at
the rear wheels of a vehicle running on E-85 than one burning gasoline only.
INCREASED ENGINE LIFE:
Ethanol has a very high MON octane rating, allowing engine
builders to run higher compression ratios without fears of destructive detonation. It also has
a very high Latent Heat of Vaporization, so the engine is cooled far better than one running
on gasoline. This lowers bottom end and oiling system temperatures substantially.
REDUCED EMISSIONS:
Although reducing emissions usually will not directly affect the on-
track performance of a race car, engine parts like pistons and valves tend to stay cleaner.
More importantly, there are serious health conc
erns with many of the octane boosting additives
that must be added to racing gasoline in order for them to be compatible with higher
compression racing engines. Ethanol fumes are also non-toxic.
FIRE SAFETY:
While many factors enter into the causes of fuel fires, ethanol does enjoy
certain advantages over gasoline fuels. The flame temperature for ethanol is lower, 1920°C
compared to 2030°C for gasoline. Because it contains less heat energy than gasoline per
volume, less total heat is released with ethanol when burning a given volume of fuel. The auto
ignition temperature of ethanol is lower than that of many gasoline, 360° compared to up to 460°
for gasoline. Liquid gasoline has vapors present at all temperatures, down to or exceeding -40°
C. Ethanol has little or no vapor pressure below 13° C. While pure ethanol fires have little or no
color, even slight amounts of gasoline added to denature (poison) the mix create bright colors
and dark smoke.
Why Not Methanol?
ETHANOL IS LESS CORROSIVE:
Ethanol is less corrosive to most fuel delivery system
components than methanol. This is due in part to the higher oxygen content of methanol.
However, certain materials that may be used as sealers or glue in methanol-compatible fuel
cells may not be compatible with ethanol. There are solutions to this problem that are being
developed for the IRL (Indy Racing League) series that will be running pure ethanol in the
2007 season.
ETHANOL IS NON-TOXIC:
Ethanol is really grain alcohol—the kind present in all alcoholic
beverages. In order to be legally transported, it must contain a “denaturing agent” (poison)
which is usually ordinary gasoline. By comparison, methanol (wood alcohol) is toxic and is
harmful or fatal if swallowed. When blended with gasoline, methanol releases formaldehyde in
passenger cars until the catalytic converter warms up. No auto manufacturer recommends
concentrations of methanol higher than 5% in their cars. However, all car manufacturers have
approved ethanol-enhanced gasoline, up to 85% ethanol content in “Flexible Fuel Vehicles”.
What is Required to Run Ethano
l Fuels in a Racing Application?
COMPATIBLE FUEL SYSTEM COMPONENTS:
Because ethanol contains oxygen, it can
form corrosive agents. Any water that enters the
system can promote the formation of formic
acid. Although this process takes a significant amount of time before damage occurs, the tank,
pump, and lines should be either stainless steel or coated with a plastic material that is
ethanol-compatible. All natural rubber parts that could be in contact with ethanol must also be
replaced with synthetic and other materials. These are all readily available from manufacturers
of racing carburetors and racing fuel system components. In some cases it may be desirable to
modify the carburetor so that it can handle the required increased liquid fuel flow when
converting from a gasoline application.
When considering fuel pump compatibility, it must also be understood that gasoline is an
insulator but ethanol does conduct electricity. This should not be an issue for race cars, as
they nearly always have a pump that is mounted
outside of the tank. However, the pump must
be internally ethanol/methanol compatible and must be able to keep up with the increased fuel
delivery rates. For information on mechanical pumps, you will have to contact the manufacturer
to be sure the pump you plan to run is alcohol/ethanol compatible.
MANAGING FUEL VAPOR PRESSURE:
Ethanol does contribute to increased vapor
pressures when the underhood temperatures ar
e very high. This issue should be easily
resolved if it becomes a problem by adding a cooler to the fuel lines.
STORING ETHANOL-ENHANCED FUELS:
All alcohols attract water and should therefore
be stored in UL-approved fuel containers that are ethanol-compatible and that limit exposure to
outside air. However, it takes a very large amount of water in the fuel (about 4 tsp. per gallon)
before “phase separation” between the mixed gasoline and ethanol occur. Water in smaller
amounts than that will result in phase separation of pure gasoline.link to the article can be found here http://iqlearningsystems.com/ethanol...cteristics.pdf
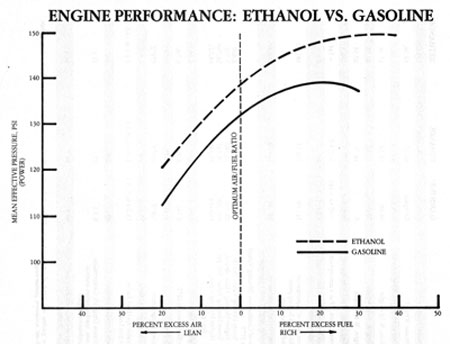
The Hot and Cold of E85 vs Gas: A Theoretical Analysis
The general consensus is ethanol burns “cooler” than gas. Technically that is correct but what does that really mean? There’s a lot more to it and I’m throwing out my analysis (btw, this is not copied from any textbook or external resource--just my own back-of-the-envelope calcs so I make no assurances).
The burning question in my mind has been how does cooler burning correlate to cylinder temps and power? That is, how can a fuel that burns “cooler” generate more power? How much “cooler” does it burn? Are the EGTs cooler? To explain this, I did some calcs--the results seem reasonable so I thought I would post them here with explanation.
If you take one mole (6.02 x 10^23 molecules) of octane (gasoline) and one mole of ethanol and combust them in air, octane would produce 5460 KJ of heat compared to 1368 KJ for ethanol (eqs 1 and 2). The heat given off from the combustion reaction can be experimentally measured to a very precise number. In this case, more heat is evolved from combusting one mole of octane than one mole of ethanol, in fact, about 4 times as much.
C8H18 + 12.5O2 → 8CO2 + 9H2O DH (heat released) = -5460 KJ/mol [eq 1]
C2H5OH + 3O2 → 2CO2 + 3H2O DH = -1368 KJ/mol [eq 2]
(note: negative sign just means heat is released; more negative = more heat)
So in this sense, ethanol burns cooler.
Now, let’s take a look at what happens inside the cylinder when the oxygen content is the limiting factor. For this, we need to look at the combustion equations where the oxygen content is set as the limiting reagent (i.e. 12.5 moles). For every 12.5 moles of O2, you can burn 1 mole of octane and 4.16 moles of ethanol. So you can see in this case, you actually get more heat from ethanol per unit oxygen (approximately 4.4% more) [eq 3].
C8H18 + 12.5O2 → 8CO2 + 9H2O DH (heat released) = -5460 KJ/mol
4.16C2H5OH + 12.5O2 → 8.3 CO2 + 12.5 H2O DH = -5700 KJ/mol [eq 3]
If we scale down the last equation by 4.4% such that the thermal energy produced from burning ethanol equals that of octane, the equation becomes
3.98 C2H5OH + 11.97 O2 → 7.95CO2 + 11.97 H2O DH = -5700/1.044 = -5460 KJ/mole [eq 4]
So how does ethanol burn cooler, yet produce more power? Well, power is a result of cylinder pressure. For that, we need to take into consideration the total number of moles of combustion products from octane and ethanol. For octane, you get a total of 17 moles of combustion products (eq. 1). For ethanol, you get a total of approximately 20 moles of combustion products (eq 4). That corresponds to approximately 18% more moles of exhaust products from burning ethanol at the same thermal energy level as octane.
Since P=nRT/V, pressure is proportional to not only T (temperature) but also n (# of moles of total exhaust products) at constant V. Well, if EtOH produces 18% more moles of gas at the same thermal energy level as octane, then the temp can drop by 18% to produce the same cylinder pressure. Hence, ethanol can burn cooler to give the same pressure as burning octane because it produces a greater amount of combustion products.
Here’s the rub--everyone wants max power, so you end up burning as much ethanol as there is O2 in the cylinder which thereby produces 4.4% more heat than octane with even greater cylinder pressures. So while ethanol can burn cooler and produce more power, in reality, we end up burning as much ethanol as possible to get max power. This results in higher cylinder temps than gas (but more power too).
e85 burns at a lower temperature than 93 octane fuel does and this will help with reducing heat spots in the combustion chamber, that will ignite the fuel prematurely. also because you are dumping so much more fuel in the intake port it will actually cool the intake air charge. additionally e85 has a much higher auto ignition temperature than 93 octane as well. whcih means that it requires a much hotter hot spot in the combustion chamber to pre ignite. for this reason you can run alot more timing on e85 and get away with higher cylinder presures than with 93 octane. but if the conditions are right there will be pre ignition which could result in engine damage.
E85 PRO’S
- It’s a renewable fuel. Unlike the fossil fuels we are so dependent on, E85 is derived from starch crops like corn that can be quickly replenished.
- Less foreign dependency. As stated before, America is heavily reliant on other countries for the source of our fuel. Generating our own fuel supply would make us a more self-sufficient nation. This isn’t a new concept, as countries like Brazil and Sweden have answered their oil crises in this way.
- Higher detonation temperature. At a combustion temp of 689 F, this fuel source is far less likely to prematurely detonate in your engine’s combustion chamber. This is partly what makes E85 so popular among racing enthusiasts.
- It’s cleaner burning. Unlike the billows of black smoke you see coming from old exhausts, E85 is a cleaner burning fuel. This minimizes harmful deposits that can build up in your engine in addition to reducing outdoor car smog.
- Cooler intake charge. Another benefit that appeals to gear heads is E85’s cooling effect. Ethanol is a denatured alcohol that creates a slight cooling effect when passing through the intake tract. This ultimately leads to increased performance.
- It’s potentially damaging. E85 is not corrosive in nature, but does attract moisture; and water is bad news for engines. It can also damage rubber seals, gaskets, and other non-metallic components.
- It can be costly to retrofit. Vehicles produced before 1987 are not E85 compatible. Many, if not all, fuel system components will have to be replaced to transform most vehicles to a flex fuel system.
- Not quite environmentally friendly. While ethanol does burn cleaner than gas, meaning less smog, flex fuel cars still produce roughly the same amount of greenhouse gases as their fossil fuel counterparts.
- Not as efficient. A car running on E85 has about 80% the efficiency of the same car running on gasoline. While you might be saving at the pump, you’re probably coming back to top off your tank more often.
- Bad in cold weather. E85 got its name because it is 85% ethanol mixed with 15% gasoline. It’s not 100% ethanol because the gasoline helps the ethanol ignite at lower temps. Colder climates may actually require E70 or lower because the high ignition temperature of the ethanol will keep your car from starting in a blizzard.
Last edited by a moderator:
NOT A TA
reliable source of info
Chemical engineer for gas company told me shelf like of modern ethanol laced gas is about 8 weeks. If you're going to keep it longer use an additive like Grumpy suggests. Old gas (but not really old) can be diluted with new gas & burned up a little at a time in a daily driver. Fill the tank, drive out a couple gallons and put in a couple gallons of the old stuff to top it off, then run it down to almost empty & repeat.
if your cars sitting for weeks or months at a time use a fuel stabilizer,
this can prevent a host of serious but avoidable issues later.
allowing a car to sit for months with ethanol laced fuel , thats not treated with stabil or something similar ,
is a sure recipe for eventual problems, adding several ounces of MMO to each tank fill also helps
http://pure-gas.org/extensions/map.html
https://gasnearmestation.com/non-ethanol-gas-near-me/
E85 gas station locations
http://e85locator.net/E85locations.html
ethanol in fuel absorbs moisture from outside air over time,
moisture causes rust


https://petroclear.com/resources/dont-be-phased.php
https://www.amazon.com/STA-BIL-2224...0&qid=1557935392&s=gateway&sr=8-1-spons&psc=1
https://www.westmarine.com/WestAdvisor/Busting-Ethanol-Fuel-Myths
http://www.lcbamarketing.com/phase_separation_in_ethanol_blen.htm
this can prevent a host of serious but avoidable issues later.
allowing a car to sit for months with ethanol laced fuel , thats not treated with stabil or something similar ,
is a sure recipe for eventual problems, adding several ounces of MMO to each tank fill also helps
http://pure-gas.org/extensions/map.html
https://gasnearmestation.com/non-ethanol-gas-near-me/
E85 gas station locations
http://e85locator.net/E85locations.html
ethanol in fuel absorbs moisture from outside air over time,
moisture causes rust


https://petroclear.com/resources/dont-be-phased.php
https://www.amazon.com/STA-BIL-2224...0&qid=1557935392&s=gateway&sr=8-1-spons&psc=1
https://www.westmarine.com/WestAdvisor/Busting-Ethanol-Fuel-Myths
http://www.lcbamarketing.com/phase_separation_in_ethanol_blen.htm
Last edited:
Maniacmechanic1
solid fixture here in the forum
MMO Marvel Mystery Oil really does work !
Unforgiven
solid fixture here in the forum
mixing old gas with chunks of wood circled by about a dozen or so rocks really creates a great atmosphere in the late evening!

